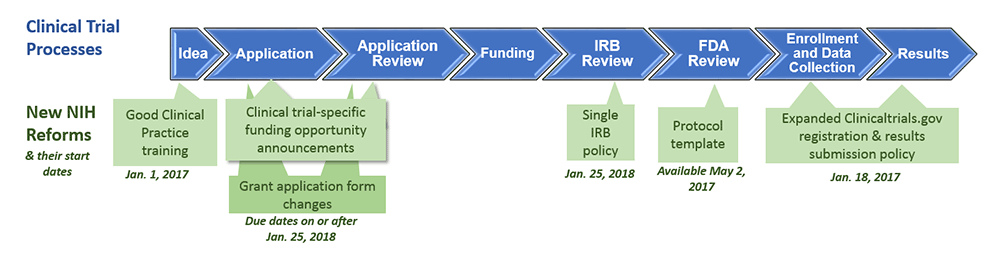
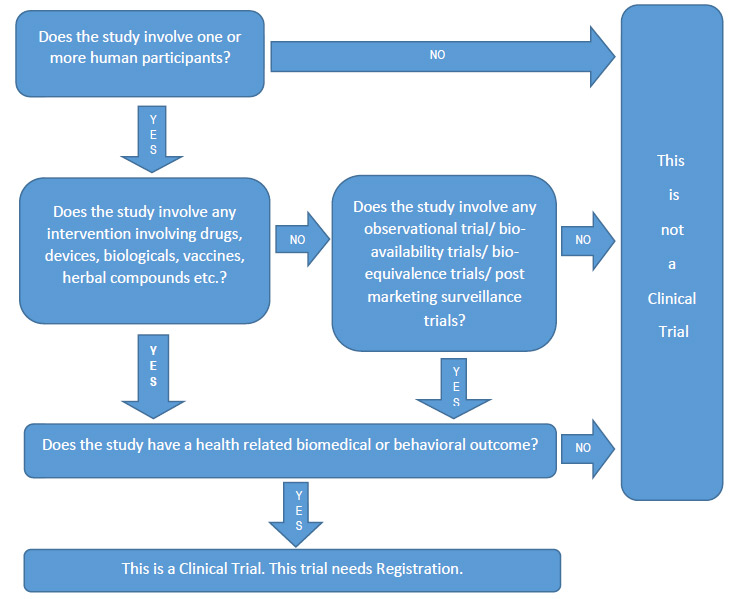
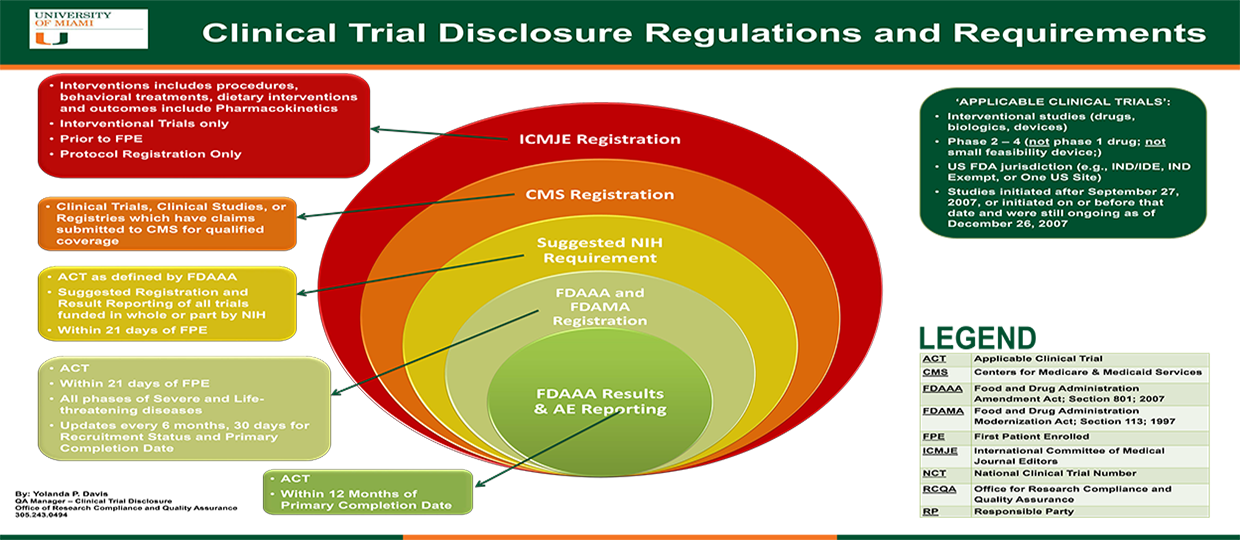
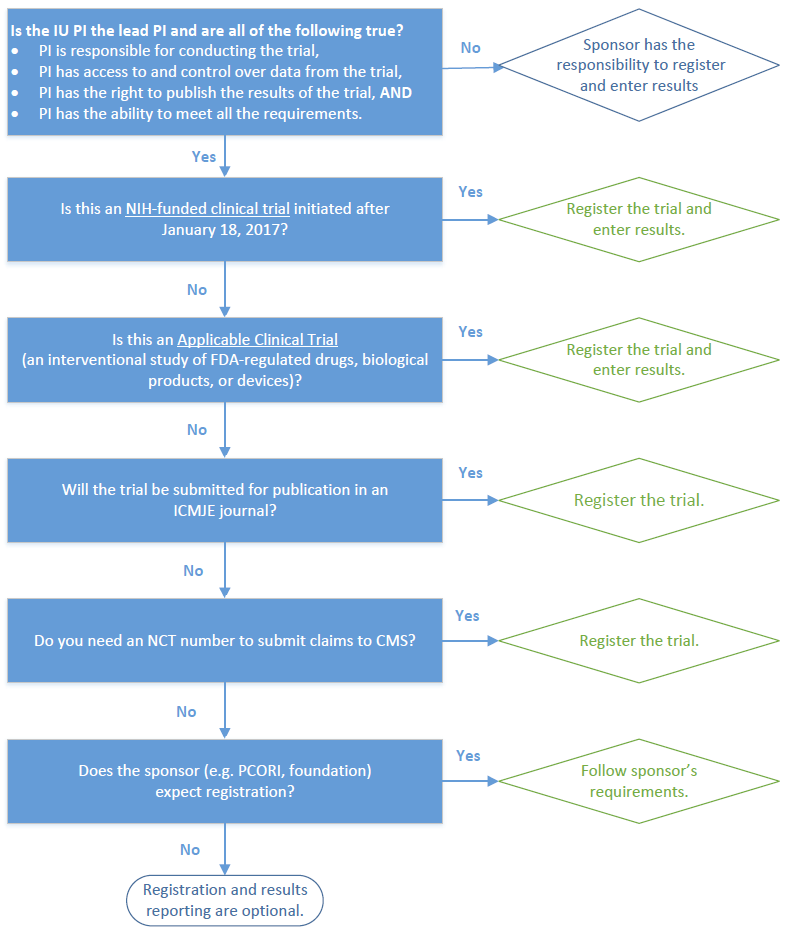
The evolution in registration of clinical trials: a chronicle of the historical calls and current initiatives promoting transparency | Semantic Scholar

Clinical trial registration: a statement from the International Committee of Medical Journal Editors - The Lancet

Clinical trial registration and publication along the drug development... | Download Scientific Diagram

Compliance with legal requirement to report clinical trial results on ClinicalTrials.gov: a cohort study - The Lancet